Our Focus
At Cortalix we are developing an internal and growing Cortalix VHH pipeline of radiopharmaceuticals, CAR-T immunotherapeutics and bioanalytical tools based on VHH antibodies. Our internal programs largely consist of radiopharmaceuticals for fibrogenesis-related membrane receptors and proteins that occur in early-stage fibrotic diseases, such as pulmonary fibrosis, cardiac fibrosis and liver fibrosis, but also in various solid cancers with fibrotic stroma, such as colon cancer and pancreatic cancer.
Our Internal Programs
The current Cortalix VHH pipeline includes internal programs in radiopharmaceuticals and CAR-T immunotherapies targeting PDGFRA (CD140a), PDGFRB (CD140b), EGFR, IGF-2R (CD222), and FAP. These programs are built on our expanded VHH discovery platform, which integrates both synthetic and immune libraries to generate high-affinity binders across diverse target classes.
A growing part of our portfolio consists of bispecific and trispecific VHH antibodies, engineered to simultaneously engage multiple disease markers or to combine target recognition with albumin binding for improved pharmacokinetics. While some of these constructs are built around our core targets such as PDGFR, EGFR and FAP, many additional targets are also included, depending on the specific application and modality. Most of our VHH constructs include a functional moiety for site-specific chelation of radionuclides such as 68-Ga for PET imaging or 177-Lu for targeted radiotherapy.
This versatile platform supports a theranostic strategy, using the same nanobody backbone for patient selection, targeted intervention, and longitudinal monitoring, all within a single molecular construct.
Our frontrunner candidate CTX001 is a PDGFRB-binding VHH antibody with excellent properties, initially developed as a 68-Ga-labeled PET imaging agent for idiopathic pulmonary fibrosis and metastatic colon cancer.
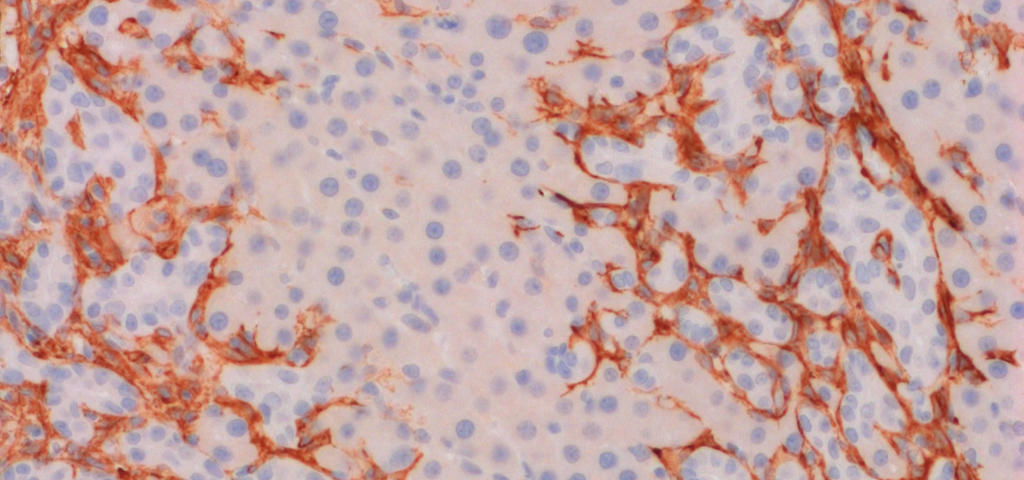
Fibrosis and fibrogenesis
Fibrosis is the consequence of chronic cycles of tissue injury and repair leading to the accumulation of scar tissue, mainly collagen, in affected organs and disruption of normal tissue architecture and function. Fibrosis is the final common pathway in virtually all forms of chronic organ failure. The cellular and molecular biology of fibrosis is similar across multiple organs and multiple sources of tissue injury, including fibrotic stroma in many solid cancers. Fibrotic stroma can make up to 80% of tumor mass and is a suitable target for cancer treatment.
Fibrogenesis is the mechanism of fibrosis and is reflecting fibrosis activity.
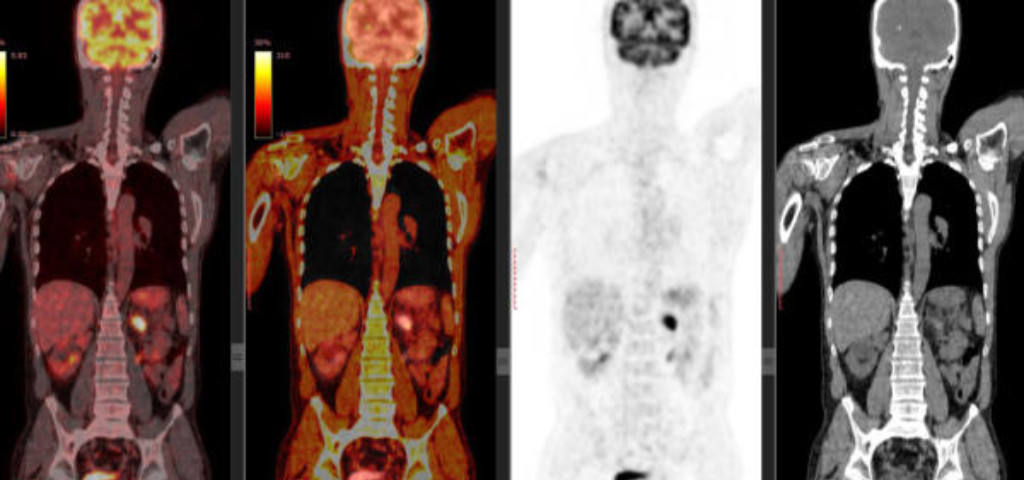
Why targeting fibrogenesis
Consequently, fibrogenesis is a far better target as it reflects fibrosis activity. Fibrogenesis is the mechanism of wound healing and repair. Upon scarring, there is migration, proliferation and differentiation of fibrogenic cells towards activated myofibroblasts, the key pathogenic cells, followed by deposition of extracellular matrix proteins, including collagen. PDGF and TGF-beta are the main drivers of fibrosis and are validated targets for fibrosis treatment. Activated myofibroblasts express specific types of membrane receptors that distinguish them from quiescent fibroblasts.
When targeting fibrogenesis, one targets the pathogenic cells of the active disease itself. Irrespective of the amounts of deposited collagen in the fibrotic organ and how fast it will disappear.
This is the area where Cortalix, as a fibrogenesis targeting company, has made fast progress in developing tracers for these specific membrane receptors, reflecting fibrosis activity.
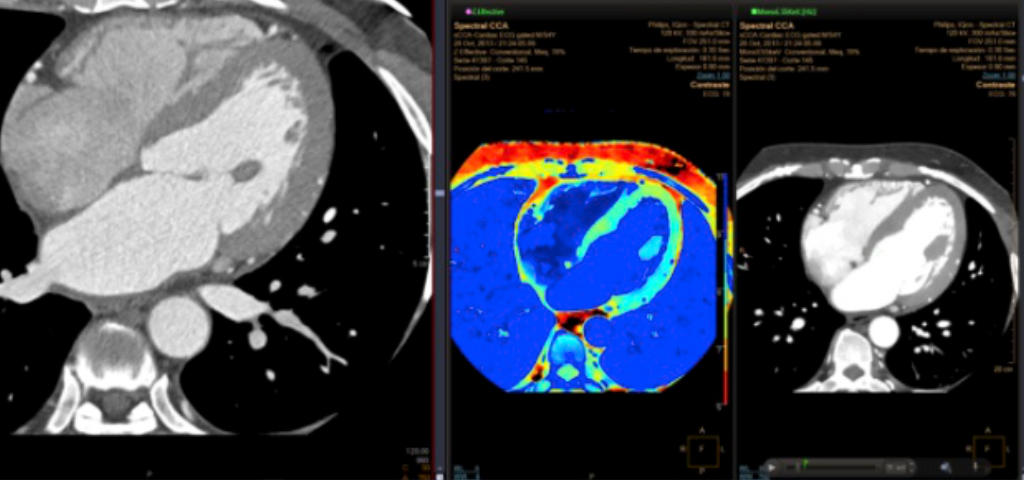
Diagnostic imaging of fibrogenesis
Diagnostic imaging of fibrosis is nowadays primarily focusing on major tissue density differences caused by scar tissue, mainly collagen. This could work for identification of patients with established fibrosis. But resolution of scar tissue after a potentially successful antifibrotic intervention may take many years. Therefore, todays’ diagnostic imaging of fibrosis is not suitable for screening and disease monitoring in patients. Moreover, lack of sensitive and reliable diagnostic options hamper the development of novel antifibrotic therapies. There is a growing demand for molecular imaging diagnostics allowing early-stage detection of fibrotic chronic diseases and help minimize costs of treating these diseases.
CTX001
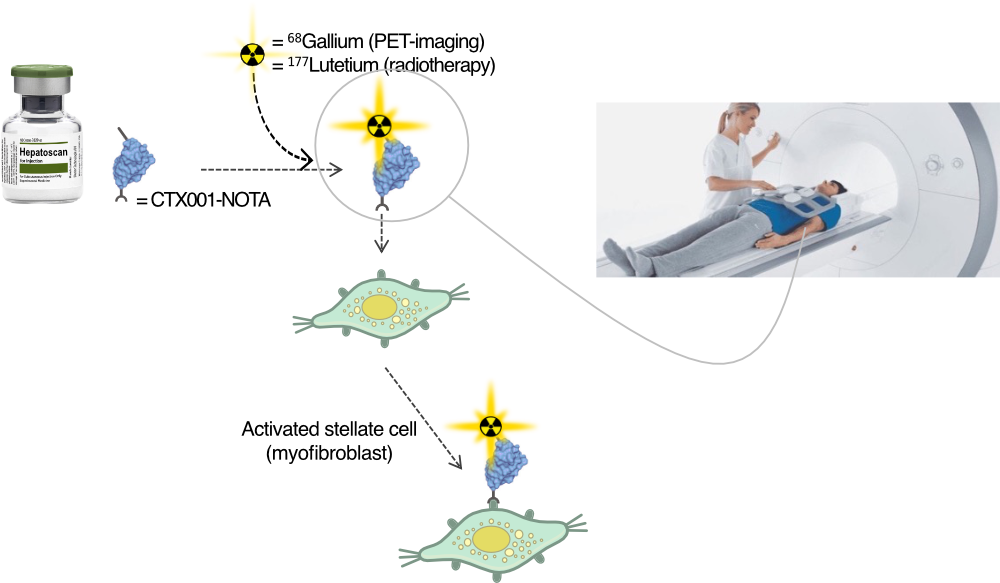
CTX001 is Cortalix’s lead diagnostic and therapeutic candidate, built on a proprietary VHH antibody platform targeting PDGFRB (CD140b), a key membrane receptor highly expressed on activated myofibroblasts, which play a central role in both organ fibrosis and the tumor stroma of solid cancers such as colorectal carcinoma.
Importantly, CTX001 is not a monospecific construct: alongside the PDGFRB-binding nanobody, it incorporates a second, non-disclosed VHH to enhance target specificity, imaging contrast, and therapeutic selectivity. This bispecific format reflects Cortalix’s broader strategy to engineer next-generation VHH constructs with improved functional performance.
CTX001 has been optimized for use in both molecular imaging and targeted radiotherapy. Its imaging version is conjugated to a NOTA chelator for 68-Ga-based PET imaging, enabling precise detection of fibrotic and tumor-associated PDGFRB expression. A fluorine-18–labeled variant is also under development to support wider clinical adoption.
For therapeutic purposes, CTX001 is being developed as a 177-Lu-DOTA-labeled ImmunoRadiotherapy agent, with an initial focus on metastatic colorectal cancer, where targeting the PDGFRB-rich tumor stroma may offer new opportunities for microenvironment-directed treatment.
BOT5035 (Thorascan)
BOT5035 (Thorascan®) was a first-generation diagnostic imaging agent based on a dimeric bicyclic peptide targeting the PDGF-β receptor. In its original format, BOT5035 was conjugated to a NOTA chelator for Gallium-68 PET imaging, with a fluorine-18 version also under development. The compound was evaluated for disease monitoring in cardiac fibrosis (including HFpEF and atrial fibrillation), pulmonary fibrosis (ILD and IPF), and liver fibrosis.
The BOT5035 program was acquired from BiOrion Technologies and progressed through multiple clinical PET imaging studies. While the compound showed promising biological targeting, its peptide-based architecture ultimately limited its further development potential.
Importantly, BOT5035 served as a blueprint for the design of next-generation nanobody-based tracers, influencing the development of VHH constructs like CTX001 and CTX004. Given the superior pharmacological properties and modularity of nanobodies, Cortalix has discontinued BOT5035 in favor of advancing its nanobody platform for both diagnostic and therapeutic applications.
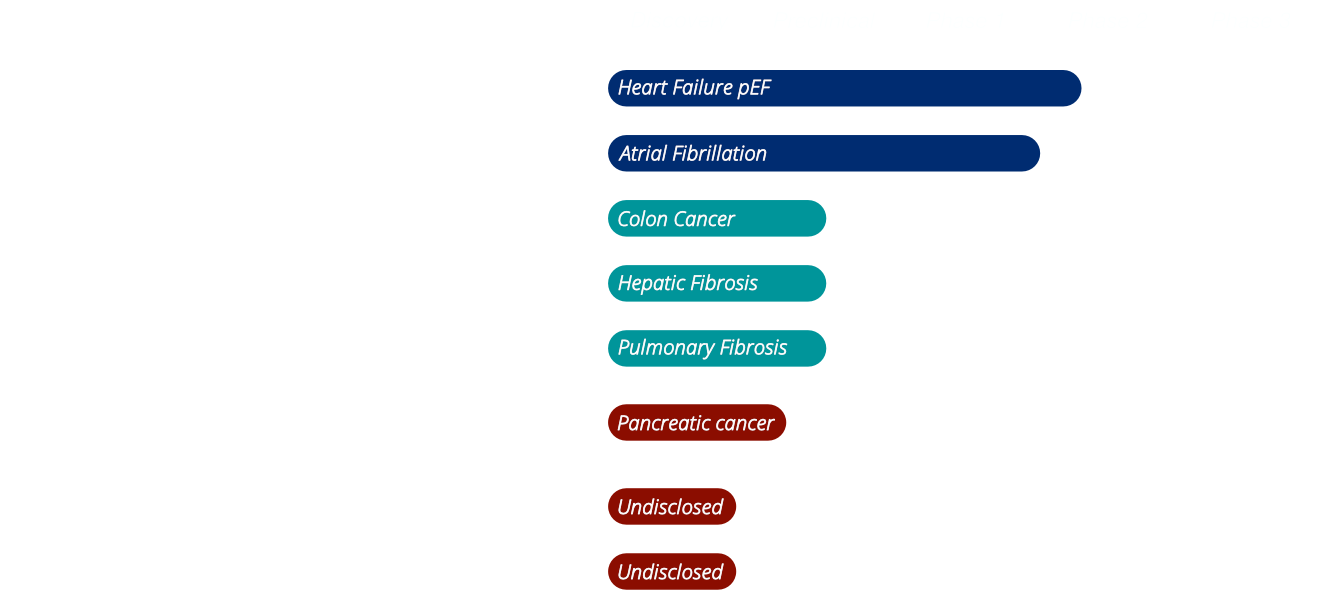
Other Candidates
In addition to CTX001, we are advancing a series of next-generation diagnostic imaging and radiotherapy candidates developed through our VHH discovery platform, targeting PDGFRA, IGF-2R, EGFR, FAP, and additional undisclosed markers. These constructs increasingly leverage our expertise in bispecific and trispecific VHH formats, designed to enhance targeting precision, signal clarity, and in vivo stability.
Our upcoming candidate CTX004, for example, is more than a PDGFRA-targeting agent. While PDGFRA remains a central component, particularly relevant for identifying patients with PDGFRA-positive pancreatic ductal adenocarcinoma (PDAC), this construct integrates a second, undisclosed nanobody specificity, tailored to strengthen both diagnostic value and clinical selectivity.
Other tracers in our pipeline follow similar principles, combining multi-target recognition with flexible labeling strategies, and will be aligned with our broader focus in fibrosis and cancer.
